Genetic Factors in GLP1 Receptor Agonist Weight Loss and Side Effects
The study explored the genetic factors influencing weight loss outcomes related to GLP-1 receptor agonist medications. Conducted among participants of 23andMe, the research analyzed the relationship between genetic predispositions and treatment efficacy, specifically focusing on weight loss and side effects associated with these medications.
Study Overview and Recruitment
The recruitment process involved individuals from the 23andMe platform. These participants voluntarily consented to share their health information for research purposes, adhering to protocols approved by the Salus Institutional Review Board. The primary goal was to gather data concerning their experiences with GLP-1 receptor agonists, initiated through a dedicated survey launched in August 2024.
Survey Details
The survey targeted participants who reported having used prescription medications for weight loss. Questions centered on:
- Drug brand and dosage
- Duration of treatment
- Weight before and after treatment
- Side effects experienced
- Reasons for continuing or discontinuing treatment
Six medications were examined: Ozempic, Wegovy, compounded semaglutide, Mounjaro, Zepbound, and compounded tirzepatide. The analysis focused on the longest-term medication for participants using multiple GLP-1 medications.
Defining Efficacy and Side Effects
The efficacy of the medications was quantified by calculating the change in Body Mass Index (BMI) before and after treatment. Participants had their pre-treatment and post-treatment BMIs measured to assess weight loss. Quality control measures were implemented, excluding individuals with extreme BMI values or those under 18 years.
Phenotype Definitions
Seats of phenotypes were established to evaluate both efficacy and side effects. For efficacy, the ΔBMI percentage was calculated using the formula:
$$Delta BMI% = 100 times frac{BMI_{2} – BMI_{1}}{BMI_{1}}$$
For side effects, separate case-control phenotypes were designated based on self-reported side effect severity. Participants rated their experiences, influencing how the data were analyzed.
Data Integration and Non-genetic Factors
The study utilized electronic health records (EHR) for comparison with self-reported survey data. Participants opted to share their health data via Apple HealthKit, enabling enhanced analysis of treatment outcomes.
Non-genetic Influencers of Weight Loss
Non-genetic factors affecting weight loss, such as drug type, dosage, age, and duration of treatment, were modeled to analyze their impact on BMI reduction. This complex relationship was examined using advanced statistical techniques.
Genetic Analysis and Association Testing
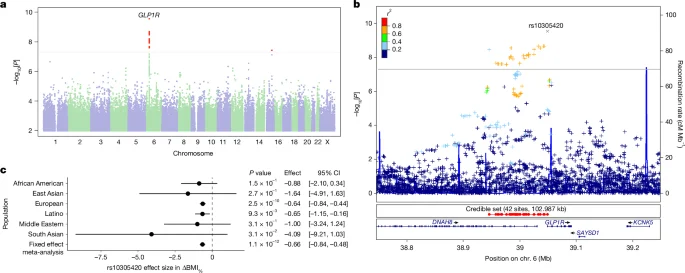
Genotyping involved analyzing saliva samples from participants, using various platforms to ensure comprehensive data collection. A genome-wide association study (GWAS) was conducted on participants of European ancestry to identify genetic variants associated with ΔBMI% outcomes.
GWAS Findings and Replication
Among 21,822 participants, analysis narrowed down to 15,237 individuals after quality filters were applied. Replication of findings was attempted in other cohorts, including the All of Us initiative and UK Biobank, though limitations were noted based on data availability.
Combining Genetic and Non-genetic Models
The study aimed to create integrated models that combine genetic and non-genetic data to better understand factors leading to weight loss and treatment side effects. These models incorporated demographic data, treatment variables, and medical histories to enhance predictive power.
Conclusion
This research sheds light on the genetic components affecting weight loss through GLP-1 receptor agonists. By analyzing both genetic and non-genetic factors, the study aims to inform future treatments for obesity and enhance personalized medicine strategies.




